
Public Education and Outreach
Coal |
Petroleum |
Natural Gas |
Oil Sands |
Oil Shale |
Biomass and BioFuels |
Hydrogen |
Nuclear |
 |
Public Education and Outreach |
|||||||
|
||||||||
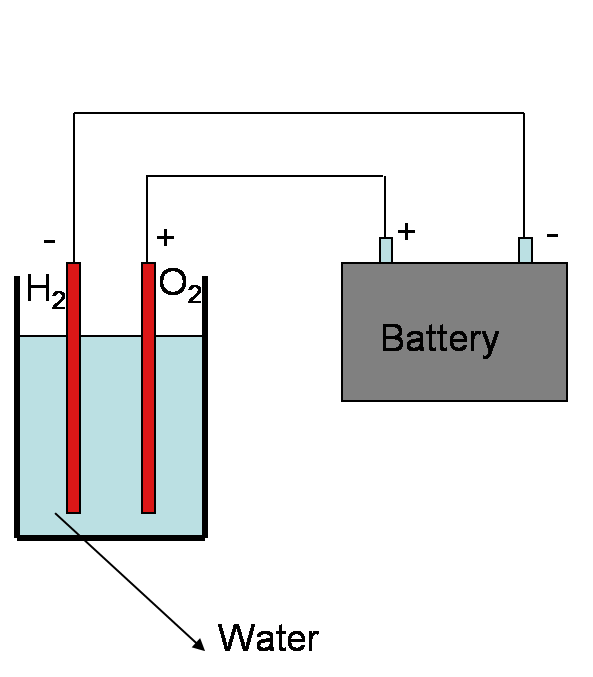
Hydrogen
Hydrogen – Where can we get?
Sequestration
Fluctuating cost of natural gas
|
 |